Introduction: Second allogeneic stem cell transplantation (SCT2) is generally associated with poorer overall survival (OS) and non-relapse mortality (NRM) than first SCT (SCT1). However, some patients survive long after SCT2, and the number of SCT2 cases increases yearly. Thus, a prognostic stratification system is required to select optimally eligible patients for SCT2. We have previously shown that the geriatric nutritional risk index (GNRI) predicts OS and NRM for SCT2 (Ann Haematol. 2020;99:1655). In this study, using a multicenter database, we aimed to validate the importance of the GNRI and establish a scoring system that can more accurately stratify the prognosis after SCT2.
Methods: We retrospectively analyzed patients with hematopoietic malignancies who underwent SCT2 between 2004 and 2020 at transplant centers of the Kanto Study Group for Cell Therapy (KSGCT), using the KSGCT database. The primary endpoint was 3-year OS. The secondary endpoints were disease-free survival (DFS), NRM, and cumulative incidence of relapse (CIR). The GNRI was defined as 1.489×albumin(g/L) + 41.7×(weight/ideal weight) (Am J Clin Nutr. 2005;82:777). We chose a GNRI cutoff value of 92.0 according to our previous report. We defined acute leukemia and malignant lymphoma in the first or second complete remission, CML in the 1st or 2nd chronic phase, and MDS with refractory anemia as standard risk, and other malignant diseases as high risk.
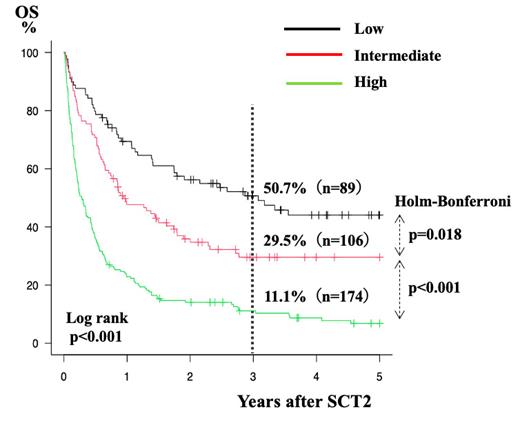
Results: Among 722 patients who underwent SCT2, we were able to calculate the GNRI and perform a detailed analysis for 376. The median patient age was 44 years (range: 16-69). The underlying diseases were AML (n=207, 55.1%), ALL/LBL (n=107, 28.5%), MDS (n=33, 8.8%), CML (n=11, 2.9%), and other (n=18, 4.8%). The median follow-up period for survivors was 1375 days (range: 223-4213). Three years after SCT2, the OS, DFS, NRM, and CIR were 26.6%, 21.7%, 40.4%, and 37.9%, respectively. Based on previous research, we divided patients into two groups: low GNRI ( ≤92) (n=162, 43.1%) and high GNRI (>92) (n=214, 56.9%). In the low GNRI group, OS was better, NRM was lower, and CIR was comparable to that in the high GNRI group (3y-OS 36.0% vs. 14.3%, p<0.001; 3y-NRM 29.5% vs. 55.2%, p<0.001; 3y-CIR 33.4% vs. 41.3%, p=0.146). Through univariate analysis and stepwise multivariate regressions on the dependent variables for OS, low GNRI (hazard ratio [HR] 1.39, 95%CI 1.05-1.85, p=0.024), high C-reactive protein (CRP) (>0.5 mg/dl; HR 1.96, 95%CI 1.16-2.63, p<0.001), and disease status at SCT2 (HR 1.54, 95%CI 1.16-2.04, p=0.003) were associated with poor OS. Likewise, low GNRI (HR 1.71, 95%CI 1.18-2.47, p=0.0045), high CRP (HR 1.69, 95%CI 1.20-2.38, p=0.0029), high HCT-CI (≥3) (HR 1.58, 95%CI 1.12-2.23, p=0.0092) were unfavorable factors for NRM, while the shorter interval from SCT1 to SCT2 (HR 1.53, 95%CI 1.10-2.13 p=0.011) and high-risk disease status at SCT2 (HR 1.62, 95%CI 1.16-2.27, p=0.0046) were associated with high CIR. To predict OS after SCT2, we developed a scoring system consisting of three factors: low GNRI, high CRP level, and high-risk disease status. As the HRs for OS were comparable, a score of 1 was assigned to each factor. OS at 3 years was 50.7%, 29.5%, and 11.1% for patients with low (score 0, n=89), intermediate (score 1, n=106), and high (scores 2 and 3, n=174), respectively (p<0.001, Fig 1). Differences between groups were revealed using the Holm-Bonferroni correction.
Conclusion: Low GNRI, high CRP level, and high-risk disease status were significantly associated with inferior OS after SCT2. The scoring system with these factors stratified the entire cohort into three risk groups including low risk with over 50% OS, and high risk with a very limited chance of survival.
Disclosures
Najima:Chugai Pharmaceutical Co., Ltd.: Speakers Bureau; Bristol-Myers Squibb K.K.: Speakers Bureau; Amgen Inc.: Speakers Bureau; Otsuka Pharmaceutical Co., Ltd.: Speakers Bureau; AbbVie GK: Speakers Bureau; Daiichi Sankyo Co. Ltd.: Consultancy, Speakers Bureau; Sumitomo Pharma Co., Ltd.: Speakers Bureau; Astellas Pharma Inc.: Consultancy, Speakers Bureau; Takeda Pharmaceutical Company Limited.: Speakers Bureau; Novartis Pharma K.K.: Speakers Bureau; CSL Behring K.K.: Speakers Bureau; Nippon Shinyaku Co., Ltd.: Speakers Bureau; Janssen Pharmaceutical K.K.: Speakers Bureau; Kyowa Kirin Co., Ltd.: Speakers Bureau. Harada:Nippon Shinyaku: Research Funding. Tanaka:Chugai Pharmaceutical: Speakers Bureau; Daiichi Sankyo: Speakers Bureau; Kyowa-Kirin: Speakers Bureau; MSD: Speakers Bureau; Otsuka Pharmaceutical: Speakers Bureau; Pfizer: Speakers Bureau; Sumitomo Pharma: Speakers Bureau; Astellas Phrama: Speakers Bureau; Abbvie: Speakers Bureau; Asahi Kasei Pharma: Speakers Bureau. Sakaida:Pfizer: Consultancy, Speakers Bureau; Otsuka: Consultancy; Ohara: Consultancy; Human Life Cord Japan: Consultancy; Chugai: Research Funding; Kyowa-Kirin: Research Funding; Takeda: Speakers Bureau; Janssen: Speakers Bureau; Novartis: Consultancy, Speakers Bureau; Bristol Myers Squibb: Consultancy, Research Funding, Speakers Bureau. Takahashi:Daiichi Sankyo RD Novare, Otsuka Pharmaceutical: Research Funding; Nippon Sinyaku: Honoraria; Novartis: Honoraria; Chugai Pharmaceutical: Honoraria; Asahi Kasei Pharma: Honoraria; AstraZeneca: Honoraria; Terumo: Honoraria; Kyowa Kirin: Honoraria; JCR pharma: Honoraria. Yano:Astra Zeneca: Honoraria; Otsuka Pharmaceutical: Research Funding. Doki:Janssen Pharmaceutical K.K.: Honoraria; Novartis Pharma K.K.: Honoraria. Kanda:Bristol Myers Squibb: Speakers Bureau; Novartis: Speakers Bureau; Chugai Pharmaceutical: Research Funding, Speakers Bureau; Pfizer: Speakers Bureau; Sanofi: Speakers Bureau; Janssen Pharmaceutical: Speakers Bureau; Kyowa Kirin: Research Funding, Speakers Bureau; MSD: Speakers Bureau; Saitama Hokeni Kyokai: Speakers Bureau; Daiichi Sankyo: Research Funding, Speakers Bureau; Asahi Kasei Pharma: Research Funding, Speakers Bureau; Meiji Seika Pharma: Speakers Bureau; Nippon Shinyaku: Speakers Bureau; Takeda Pharmaceutical: Research Funding, Speakers Bureau; Amgen: Speakers Bureau; Sumitomo Pharma: Research Funding, Speakers Bureau; Human Life CORD: Speakers Bureau; AstraZeneca: Speakers Bureau; Otsuka Pharmaceutical: Research Funding, Speakers Bureau; Japan Blood Products Organization: Research Funding, Speakers Bureau; CSL Behring: Speakers Bureau; AbbVie: Research Funding, Speakers Bureau; Towa Pharma: Speakers Bureau; FUJIFILM Wako Pure Chemical: Speakers Bureau; Wakunaga Pharmaceutical: Speakers Bureau; Eisai: Research Funding, Speakers Bureau; Precision: Speakers Bureau; Alexion Pharma: Speakers Bureau; Shionogi Pharma: Research Funding; Taiho Pharmaceutical: Research Funding; JCR Pharmaceuticals: Research Funding; Nippon Kayaku: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal